I want to make an application to access data through Data Access Request Service (DARS)
How to request access to health and social care data through DARS for purposes intended to help improve NHS services. This guidance includes the different application stages, what information is required, and the checks we carry out before data access is approved.
Help us improve
We would like to find out how you use the DARS web pages.
How to make a new request to access data through DARS
Organisations requesting access to NHS data must be able to demonstrate that they can meet all of NHS England’s legal, ethical and security requirements for safe data use.
The steps in the DARS process ensure these requirements are met.
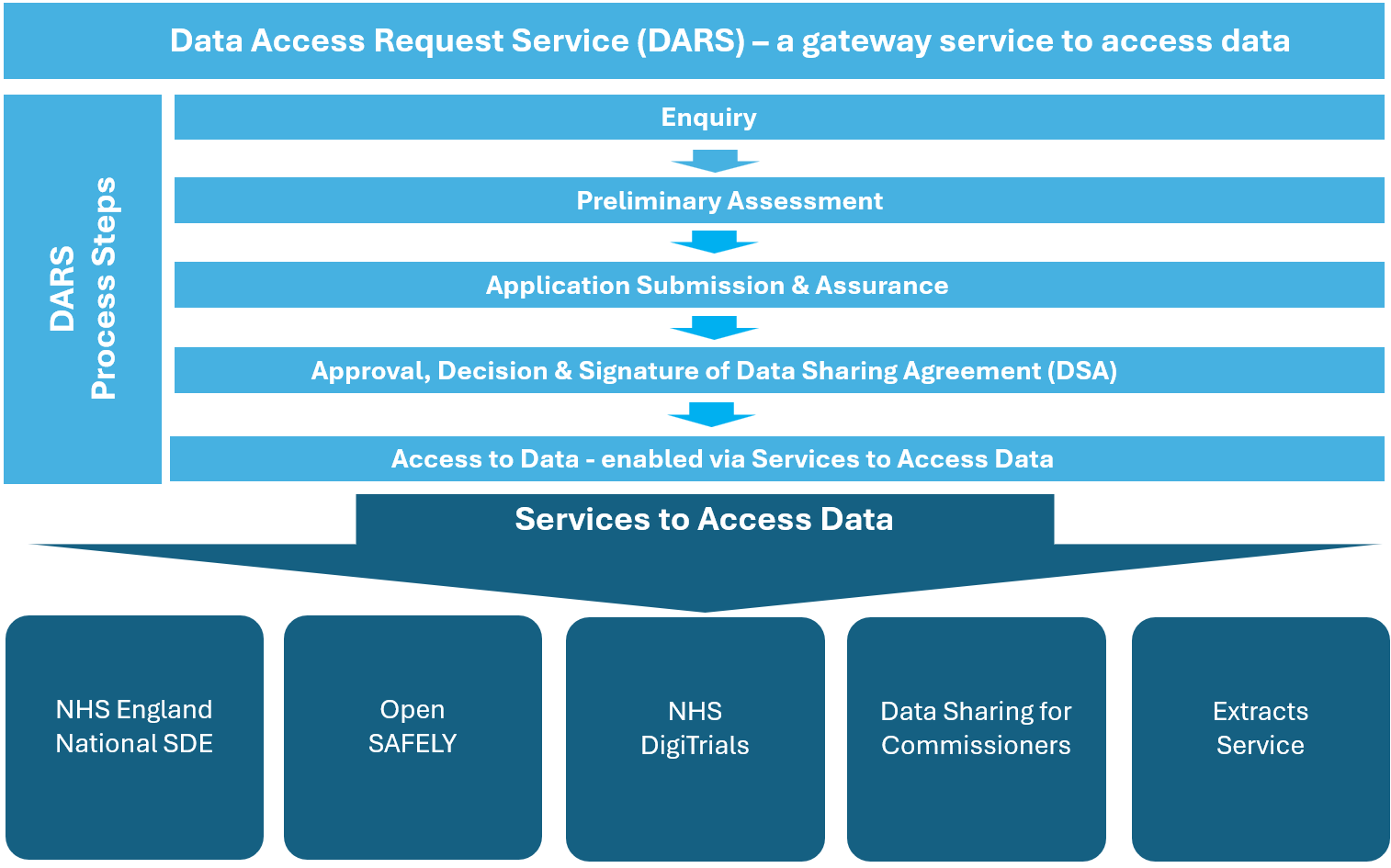
Enquiry
If you are thinking about making a request to access data using DARS, you should contact the DARS data applications team by email including the information below.
- details of what NHS England data you want to access
- summary of why you wish to request access to the data
- names of the organisations who will be involved in the data access
- an indication of your readiness to make a request access to data
We recommend making an enquiry as early as possible. For example, when you are at the early stages of applying for funding for your project, or you anticipate your request for data may be complex or novel.
Guidance on the information required in your enquiry can be found in the DARS applications supporting guidance.
We will review your enquiry to ensure that data you are requesting is available for access and technically feasible through DARS.
We may contact you to ask you to provide more information.
If your data needs can be met by DARS, we will move your enquiry to the preliminary assessment stage. During this stage we will undertake a more in depth assessment of your specific data access needs, along with the legal, ethical and security requirements, and appropriate services to access data to meet them
Preliminary assessment
The preliminary assessment looks at how DARS can meet your needs. We have provided additional guidance to support your application.
- the most suitable service to access data based on your data needs
- how organisations involved in your data access will meet any ethical, legal and the evidence you will need to provide
- any preparations needed for data access, for example, submitting details of a participant cohort to NHS England
- the relevant agreements needed for your data access request. This will ensure you can identify and prepare whoever needs to sign the agreements in line with any delegated authorities processes your organisation has in place
- how organisations involved in data access will meet any security requirements including cloud storage
- an indication of how long data access may take for your request
- the estimated cost to meet your needs both of making an application, and of the service to access data this will enable you to confirm the source of funding, and provide payment details to DARS when needed
If your proposed use of the data is novel or potentially contentious, we may use our assurance process to receive early advice during the preliminary assessment.
This will provide you with an indication of if your request is likely to be supported and of the actions necessary before you can make a formal application.
Application submission and assurance
Application submission
The DARS team will advise when you’re ready to make a formal application. You will be allocated a dedicated DARS contact who will confirm and document the details of your data access request, supporting you through submission of your application through the DARS online system.
Once an application has been submitted through the DARS online system, a review will take place to ensure that the application can be accepted. This is an important step in the process, and you may be need to amend, update or correct any information in your application to ensure it can be accepted into DARS
Once an application has been accepted, it then progresses through assurance.
If GP data is included in the application an additional review may be required by the Professional Advisory Group (PAG). PAG provides NHS England with specific advice and feedback from the GP profession.
Additional assurance may be recommended, before your application can be approved. The DARS team will support you with understanding any additional work you may need to complete.
Approval, decision and signature of the Data Sharing Agreement
Following successful assurance, approval is given and we generate a Data Sharing Agreement (DSA).
The DSA is an important document as it covers details of the data access which has been approved including:
- the specific data being shared
- the purpose of the data use
- security and confidentiality measures
- legal basis for processing
- the services to access data being used for the access
The DSA must be signed by NHS England and the data controller before access to data can start.
Learn more about the DSA and other agreement documents important for data access.
Access to data
Once a signed DSA is in place, arrangements to access data are made to use the services to access data specified in the DSA.
To access data agreed in your signed DSA you may also need to:
- where applicable, sign an additional service agreement for the service to access data you will be using. More information on service agreements can be found on contract and agreements required to access data required to access data from NHS England.
- if required, promptly send participant (cohort) data to NHS England within the required time scale. Further information can be found in the guidance for submitting participant data (cohort file).
Last edited: 2 June 2026 5:28 pm